Optimizing Silver Conductive Ink for Printed Electronics”: Synthesis, Characterization, and Performance of Silver Conductive Ink for Lock Security Keypad in Printed Circuit Applications
Sai Ko Soe, Zaw Min Hein, Phyu Phyu Thwel, Kyaw Zin Oo, Maung Aye, Ye Naing, Lin Htun, Wine Htet Soe, Thida Soe,Kyawt Kyawt Khaing, Phyu Phyu Thant, Sitt Paing Myint, Maung Maung Thant
Abstract
Conductive materials enable efficient charge transport in printed electronics, which allows the creation of flexible, lightweight, and low-cost electronic devices and plays a key role in printed circuits and sensors. Compared to other conductive inks, silver conductive ink offers superior electrical conductivity and stability, making it the most widely used material in high-performance printed electronics. In this study, reactive transparent conductive silver ink was successfully synthesized via the chemical reduction of silver nitrate and sodium acetate using Tollen’s process. Ammonium solution and formic acid served as the reducing and stabilizing agents, respectively. Treatment of silver acetate with ammonium solution facilitated the formation of stable silver nanoparticles in the solution. Five different ratios of silver acetate and ammonium solution were prepared and investigated. Silver conductive inks with high electric conductivity were obtained at a ratio of silver acetate to ammonium solution of 1:3. Additionally, the effects of sonication and centrifugal force on the size of silver particles and the conductivity of printed silver ink circuit patterns were also examined. The morphology, elemental composition, and size distribution of the silver nanoparticles were analysed using a field emission-scanning electron microscope (FE-SEM) and an Energy Dispersive X-ray system SEM microscope (EDX). The viscosity of the synthesized silver ink and the thickness of different ink layers-coated circuit patterns were also investigated. The particle size of silver nanoparticles in the silver inks was successfully reduced to 168 nm after different treatments. Furthermore, the performance of silver inks containing larger and smaller particle sizes of silver nanoparticles was compared, and the ink with the best performance was successfully used in a lock security keypad system, confirming its potential for use in printed circuits, sensors, RFID tags, flexible cables, and radio frequency systems.
Keywords -Conductive silver ink, Sintering Method, Tollen’s process, Printing electronic, Circuit Lines, Lock Security Keypad.
1. Introduction
As technology advances, the need for materials that support and encourage further technological innovation becomes imperative nowadays. Conductive inks play a prominent role in this realm by supporting the development of next-generation technologies in flexible and printable electronics (Islam et al., 2025). Conductive ink is a specialized ink that is capable of conducting electricity in a printed object (Suriati et al., 2011). Despite continuous research efforts worldwide, certain complexities in the field of conductive inks remain unresolved. These include optimizing the balance between conductivity, ink stability, and ease of application, as well as addressing the scalability of production methods for commercial use (Wang et al., 2004). Among the various types of conductive inks, such as metallic inks, graphene, and carbon nanotube inks, silver conductive ink (SCI) stands out as a preferred choice for researchers worldwide because of its superior thermal and electrical conductivity, high chemical stability, and relatively low cost (L. Yang et al., 2023). SCI holds special significance due to its versatility across diverse technological fields, particularly in electronic applications within printing technology (Grouchko et al., 2011).
The chemical reduction method is the most commonly used method for synthesizing silver nanoparticles (Ag NPs), which involves reducing silver ions (Ag+) into metallic Ag NPs using a reducing agent, often in conjunction with a stabilizing agent to prevent aggregation (Iravani et al., 2014). The retention matrix of SCI embeds and stabilizes the metallic conductive Ag NPs. This matrix is crucial because it ensures the even distribution of metallic Ag NPs throughout the SCI and keeps them suspended until the ink is applied to a surface. This process maintains consistent conductivity and uniform printing quality. To create a conductive pathway with NPs in SCI, the matrix volume must be reduced through a curing or evaporation process, known as sintering. Organic dispersants and stabilizers are added to the SCI to prevent agglomeration caused by the high surface energy of Ag NPs. These organic agents decompose and allow NPs to interact more effectively during sintering. Among various reducing and stabilizing agents, ammonium solution and formic acid are particularly effective for synthesizing Ag NPs.
Ammonium solution aids in the reduction process of Ag salts to NPs by controlling the reduction rate and maintaining a stable pH of the solution. It helps to prevent agglomeration and ensures uniform size and dispersion of Ag NPs. Similarly, formic acid acts as a reducing agent, converting Ag ions into NPs through a mild reduction process. It also functions as a stabilizer by capping the surface of Ag NPs, preventing aggregation and ensuring long-term stability of the ink formulation.
Sonication parameters such as frequency, power, and duration, along with rotational speed during synthesis, significantly influence nanoparticle size and uniformity. By adjusting these parameters, particle size can be controlled, resulting in smaller, more uniform nanoparticles, which improve dispersion and prevent aggregation. Switching from liquid ink to a solid typically involves processes like drying, curing, or melting (Jiang et al., 2021). Curing temperature plays a key role in the conductivity of SCI, with an optimal range generally between 120°C and 150°C. Within this range, Ag NPs sinter effectively, enhancing electrical connectivity without causing excessive agglomeration or degrading ink stability (Dearden et al., 2005).
In this study, SCI were synthesized using Tollen’s process with some refinement. Five different ratios of silver acetate to ammonium solution were used to determine the optimal formulation for achieving the best electrical conductivity and stability. The most effective ink was then applied in making a security lock keypad using an acrylic plate as a substrate to demonstrate its potential for practical applications in electronic devices. Silver acetate was used as a precursor to synthesize SCI, providing a stable source of Ag+ that can be reduced to form Ag NPs during the ink preparation process. Ammonium solution and formic acid were used as reducing and stabilizing agents. Ammonium solution helps to reduce silver salts into Ag NPs while controlling the pH and preventing agglomeration. Formic acid not only acts as a reducing agent, facilitating the conversion of Ag+ into Ag NPs, but also stabilizes the particles by capping their surfaces, ensuring a well-dispersed and stable ink formulation. Low curing temperature at 100 °C was used to improve the electrical connectivity of the ink without causing excessive agglomeration. The effect of sonication and rotational speed on the size and uniformity of NPs was also investigated. Different characterization techniques, such as FE-SEM and EDX, were employed to analyze their morphological features and particle sizes as well, and viscosity and thickness measurements were also done to evaluate the dispersion, stability, and printability of as-synthesized SCI.
2. Materials And Methods
Materials
Silver nitrate (AgNO3, A.C.S. reagent) and sodium acetate (C2H3NaO2) were purchased from Loborat Fine Chemical Ltd.India. Ammonium Solution (NH3) and Formic Acid (CH2O2) were purchased from Qualikems Fine ChemPvt. Ltd.India. All chemical reagents were analytically pure, and no further purification was made. Deionized water (DI) was used during the whole experimental process.
Preparation of Conductive Silver Ink
Reactive transparent SCI was synthesized utilizing Tollen’s process with specific modifications. Initially, 10 g of AgNO3 was dissolved in 100 mL of DI water in one beaker, while 5 g of C2H3NaO2 was dissolved separately in another beaker containing 100 mL of DI water. These solutions were gradually mixed in a suitable beaker, resulting in the formation of a solution with white sediments. Subsequently, the solution underwent filtration using a Ф125 mm filter paper, yielding 8.49 g of silver acetate (a semi-product). Silver acetate was then combined with aqueous ammonium solution, and formic acid was incrementally added dropwise to the reaction mixture. The color changes of the reaction mixture from light orange to brown and ultimately to grey indicated the rapid reduction of silver ions to silver particles, which were subsequently filtered using a 0.45 µm syringe filter. For the preparation of five different ratios of SCI, the fixed amounts of 1 g of silver acetate, which was mixed with ammonium solution in ratios of 1:1, 1:2, 1:3, 1:4, and 1:5. The mixture was continuously stirred for 15 minutes, with formic acid added dropwise: 0.1 mL for the 1:1 ratio, 0.2 mL for the 1:2 ratio, and so on. The five samples were then placed in closed containers and left to rest overnight at room temperature for complete reaction and stabilization of the as-synthesized inks.
Studying the Effect of Sonication and Rotational Speed
In this study, the 1:3 ratio sample was selected to investigate the effects of sonication and rotational speed on the nanoparticle size due to its good electrical conductivity and ink stability than other ratio samples. To assess the impact of sonication on nanoparticle size, 20 Hz sonication was applied after adding 3 g of ammonium solution to 1 g of silver acetate solution. Following this, 3 mL of formic acid was added, and the same reaction parameters were applied again. The final sample was then sent for characterization. The different reaction parameters were used as specified in Table 1, and the particle size of the samples treated under these parameters was characterized and compared.
Coating Conductive Silver Ink on the Acrylic Substrate
The substrate selection significantly affects the performance of conductive ink coatings, with surface roughness, porosity, and heat resistance being key factors. In this study, acrylic plates were chosen for their suitable surface properties, their good surface roughness, porosity, and their melting point of 160°C. A baseline measuring 1×1 cm2 was initially drawn on the acrylic plate using base paint with a Craft Cutting Plotter (DC 240, China), which served as the foundation for subsequent ink coatings. The conductive silver ink was then applied onto the baseline and subjected to heating at 100°C using a 1000 W halogen light bulb until the ink coating was fully dried. Subsequently, the plate was allowed to cool at room temperature for approximately three minutes. To assess the best performance of the coated ink, the conductive silver ink was applied six times onto the base paint line, with the same steps followed for each coating. The thickness and electrical conductivity were measured after each coating. Furthermore, to assess the practical use of the SCI, a simple circuit was fabricated on the acrylic plate using the ink. The circuit was connected to a 5V power supply and tested with an LED light bulb. The successful illumination of the LED light bulb demonstrated the efficacy of the as-synthesized SCI in functioning within regular electrical circuit lines and patterns.
Application of Conductive Silver Ink in Making a Lock Security Keypad
A security lock keypad board measuring (9×11 cm2) was designed using SolidWorks software, providing precise dimensions and layout for the circuitry. Subsequently, the circuit lines were drawn on the acrylic substrate using a Craft Cutting Plotter to get base-keypad lines. The keypad lines were 2 mm wide and had a thickness ranging from 5 to 6 µm. After drawing the keypad lines on the acrylic plate, conductive ink was applied to the keypad circuit lines and heated at 100°C using a 1000 W halogen light bulb until the ink coating was fully dried. Subsequently, the plate was then cooled at room temperature for 3 minutes. The six coating layers of ink were applied to the acrylic plate, and its electrical resistance and conductivity were measured using a digital multimeter device to achieve the desired electrical resistance and high electrical conductivity. The functionality of the security keypad system was verified using Arduino software, ensuring its compatibility with electronic systems.
3. Characterizations
4. Results And Discussions
Analysis of EDX and FE-SEM Images
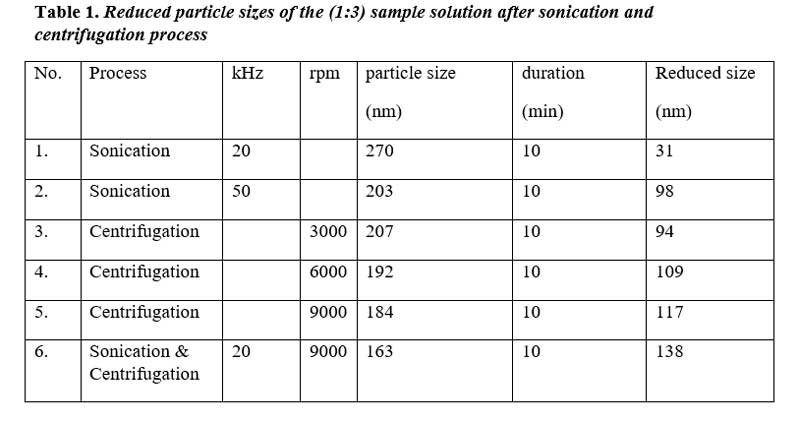
The EDX analysis of the 1:3 ratio synthesized sample confirmed a high silver content of 93.59 wt%, with minor amounts of oxygen (5.10 wt%), sodium (0.94 wt%), and magnesium (0.37 wt%) (Figure 1C). FE-SEM analysis image revealed a densely packed and relatively uniform granular surface, indicative of good particle interconnection, which is essential for maintaining continuous conductive pathways (Figure 1B). The observed particle size was approximately 301 nm, with a broader distribution visible throughout the field.
Figure 1. (a) Different ratios of conductive silver ink. (b) SEM image of (1:3) sample solution of particle size 301 nm. (c) EDX image of (1:3) sample solution.
Effect of Sonication and Rotational Speed on Particle Size
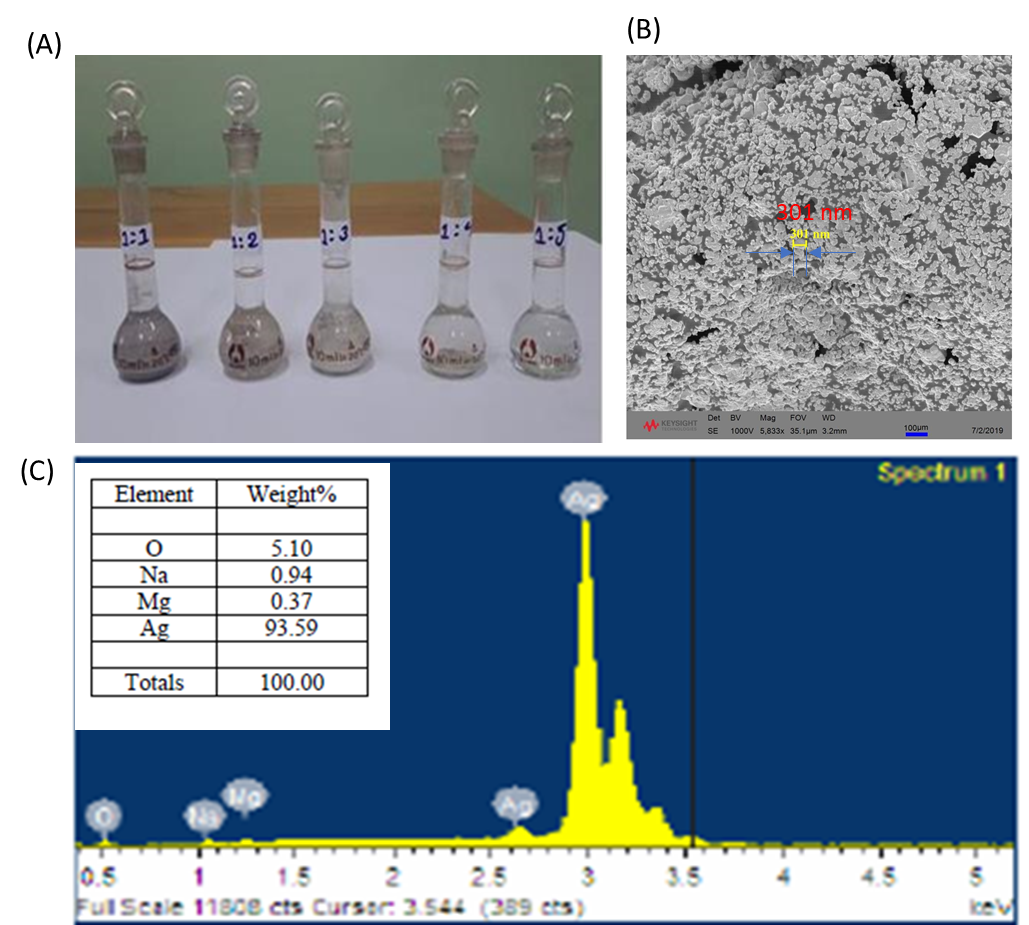
In this study, the effects of sonication and centrifugal force on particle size reduction were investigated to optimize the properties of the conductive ink. Table 1 summarizes the results obtained from these experiments. Specifically, 10 ml of silver conductive ink underwent sonication at frequencies of 20 kHz and 50 kHz for 10 minutes,150 W. The resulting particle sizes were measured at 270 nm and 203 nm, respectively (Figure 2A, B). Furthermore, centrifugation experiments were conducted on 10 ml of silver ink at different rotational speeds (3000 rpm, 6000 rpm, and 9000 rpm) for 10 minutes each. The particle sizes decreased to 207 nm, 192 nm, and 184 nm, respectively, as depicted in (Figure 2C, D, E). Additionally, combining ultrasonic homogenization at 20 Hz with centrifugation at 9000 rpm further reduced the particle size to 168 nm (Figure 2F). These findings demonstrate the significant efficacy of sonication and centrifugal force in refining the particle size distribution of the synthesized silver nanoparticles, thereby enhancing the electrical conductivity of the ink.
Figure 2. SEM images of (a) 20 kHz sonication, (b) 50 kHz sonication, (c) 3000 rpm centrifugation, (d) 6000 rpm centrifugation, (e) 9000 rpm centrifugation, and (f) 20 kHz sonication and 9000 rpm centrifugation.

Thickness Measurement
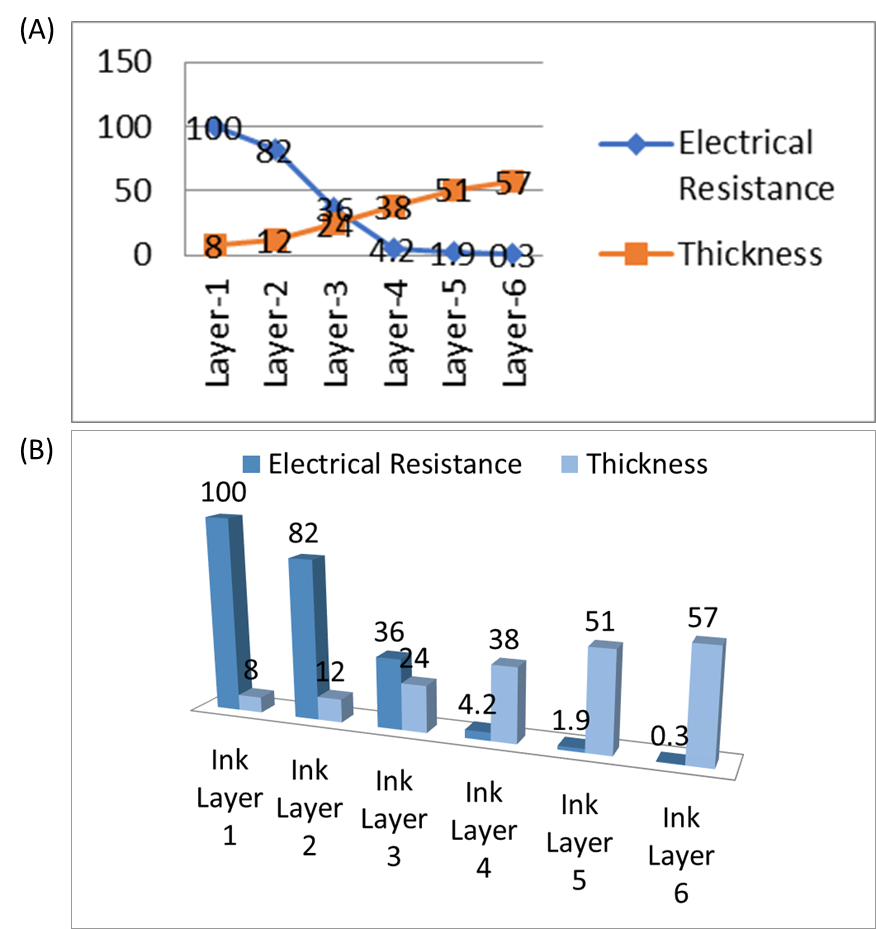
The thickness of the plane acrylic plate was 1939 µm. When a base paint layer was applied onto the acrylic plate, the thickness increased to 1947 µm, indicating a nominal thickness of 8 µm for the net ink layer. Subsequent layers of conductive ink were applied onto the acrylic plate, with varying numbers of layers to assess the impact on thickness and conductivity. The addition of one layer of conductive ink resulted in a further increase in thickness to 1959 µm, while three layers led to a thickness of 1983 µm. Notably, the application of six layers of conductive ink resulted in a total thickness of 2129 µm, indicating the increment of buildup ink layers on the substrate surface (Figure 3A, B).
Figure 3. (a) Thickness of acrylic film 1939 µm. (b) Thickness of six times ink coating on acrylic plate. (c) testing the resistivity of 1x1cm2 sample. (d) conductivity test using LED. (e) Viscosity tester. (f) Test result of ink 479.5 cP.

Electrical Resistance Measurement
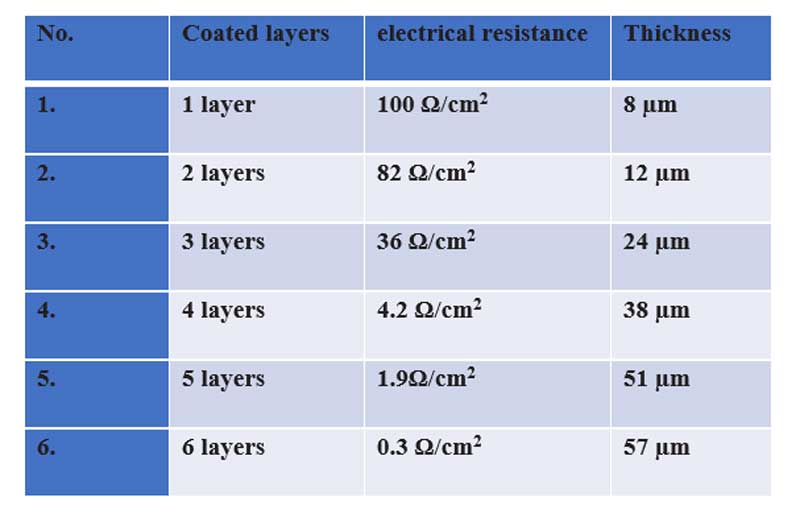
The resulting electrical resistance for different numbers of coating layers was presented in Table 2. Notably, as the number of ink layers and thus the thickness increased, the electrical resistance decreased, leading to improved conductivity of the coated ink. The correlation between thickness of net ink and its electrical resistance can be studied in Figure 4 A, B.
Figure 4. (a,b) Correlation between electrical resistance and ink thickness
Viscosity Measurement
The viscosity measurements of silver-based inks were conducted using a Visco Dens VDM-250.1 viscosity testing meter (Serial Number: 016538). The testing was performed at a temperature of 25.37°C, with a corresponding density of 1.087 g/cm3. The viscosity of the silver ink was found to be 479.5 centipoise (cP) (equivalent to 479.5 mPas) (Figure 3E, F).
Table 2. The electrical resistances and thickness of coated layers
5. Discussions
The properties of synthesized silver inks with different precursor-to-solvent ratios were evaluated to determine their suitability for use as conductive inks in printed electronics. The sample with a 1:1 ratio exhibited significant sedimentation of Ag NPs after two days, resulting in a muddy and unstable dispersion. This rapid aggregation is likely due to an insufficient solvent content to maintain colloidal stability, resulting in particle flocculation. Consequently, circuit patterns fabricated using that ink displayed poor electrical conductivity and irregular, porous surface morphology, and showed undesirable traits for reliable electronic performance. In contrast, silver inks prepared at 1:2 and 1:3 ratios demonstrated improved dispersion stability, homogeneous, and clear solutions. These ratios provided a better balance between metal content and solvent, and effective particle separation. The resulting printed patterns exhibited smooth film formation, enhanced conductivity, and lower sheet resistance, indicating optimal particle packing and improved sintering behavior. However, the 1:4 and 1:5 ratios proved unsuitable for conductive applications. The excessive solvent content reduced silver nanoparticle concentration below the percolation threshold, resulting in high resistivity and mechanically weak films. Additionally, the electrical conductivity performance from these inks was relatively low. Notably, among all tested formulations, the 1:3 silver precursor-to-solvent ratio exhibited the best overall performance, offering excellent dispersion stability, smooth film formation, and the highest electrical conductivity, making it the most suitable for conductive ink applications and was therefore chosen for further experimental investigation and application development.
EDX imaging of the (1:3) ink sample revealed a remarkable silver purity of 93.59%. This high silver content indicates minimal contamination and suggests efficient reduction and stabilization during synthesis. Such composition is highly favorable for conductive ink applications, as a high metallic silver content directly enhances the electrical conductivity of printed traces. FE-SEM imaging further identified Ag particles with an initial size of approximately 301 nm in the original (1:3) ink sample. The size of NP is critically important in conductive ink applications for several reasons. Smaller Ag NPs possess a higher surface area-to-volume ratio, which enhances their surface reactivity and facilitates better particle-to-particle contact during the film formation process. This improved connectivity between particles and then leads to denser packing and fewer voids within the printed film, thereby decreasing the overall resistivity. Furthermore, smaller particles sinter at lower temperatures, allowing for more efficient formation of conductive pathways. Therefore, we have tried to reduce the size of Ag NPs under different physiological parameters. The sonication at frequencies of 20 kHz and 50 kHz led to particle size reductions to 270 nm and 203 nm. Additionally, centrifugation at rotational speeds such as 3000 rpm, 6000 rpm, and 9000 rpm resulted in further size decreases to 207 nm, 192 nm, and 184 nm, respectively. Notably, a synergistic treatment combining 20 kHz sonication with 9000 rpm centrifugation ultimately reduced the particle size to 168 nm, demonstrating the effectiveness of this integrated approach in particle size modulation. Therefore, the combined use of sonication and centrifugation emerges as a potent strategy for tailoring nanoparticle dimensions, which in turn significantly contributes to optimizing the performance of SCI by promoting uniform dispersion, enhanced surface contact, and improved electron mobility in printed electronics.
Furthermore, the relationship between thickness of ink layer, their electrical resistivity, and conductivity has been found. The actual amount of ink deposited on the substrate is influenced by multiple factors, including the ink formulation, printing process parameters, such as speed and pressure, as well as the characteristics of the substrate material (Jiang et al., 2021). A 1×1 cm2 baseline was initially drawn on the acrylic plate using base paint. After the first layer of SCI coating, the resultant net ink thickness was 8 µm, with an electrical resistance of 100 Ω/cm2. The resistance decreased to 36 Ω/cm2 after the third coating, corresponding to an increased ink thickness of 24 µm, indicating improved electrical conductivity. Following the sixth coating, the electrical resistance further decreased to 0.3 Ω/cm² Figure 3C, with a final thickness of 57 µm, as illustrated in Table 2. This finding underscores the effectiveness of the conductive ink formulation and deposition process in maintaining electrical performance while achieving the desired ink thickness. Additionally, the correlation between ink thickness and conductivity highlights the importance of optimizing ink deposition parameters to strike a balance between ink coverage and electrical performance in practical applications. As the number of coating layers increases, there is a corresponding rise in thickness, accompanied by a reduction in electrical resistance. This phenomenon is attributed to the increase in the number of conductive layers deposited on the substrate surface. The augmentation in thickness results in a higher concentration of charge carriers available for electrical transport, consequently enhancing the overall electrical conductivity of the coated layer.
The viscosity of nano-inks is influenced by various factors, including the choice of solvents, nanoparticle dimensions, and temperature. These parameters collectively impact the flow behavior and rheological properties of the ink, thereby affecting its printability and performance. Nano-inks need to exhibit a viscosity within the range of 1-25 mPas and a surface tension between 25 and 50 mNm⁻¹ to ensure optimal printing characteristics and deposition quality. However, the viscosity of our synthesized ink was 479.5 centipoise (cP) (equivalent to 479.5 mPas). This high viscosity value exceeds the desired range for printable nano-inks, indicating a need for viscosity reduction to achieve the target viscosity range of 1-25 mPas. Therefore, we chose coating the ink on the substrate instead of directly printing. Adjusting the ink formulation or incorporating viscosity modifiers may be necessary to achieve the desired viscosity level conducive to efficient printing and performance in various applications. Finally, the practical utility of the developed conductive silver ink was demonstrated through the successful fabrication of a lock security keypad measuring 9×11 cm2. The keypad drawn using the (1:3 ratio) SCI exhibited functionality comparable to commercially available counterparts, thereby validating the efficacy and the real applicability of the synthesized ink (Figure 5).
Figure 5.
(a) Drawing the keypad using SolidWorks software. (b) Plotting the design with a cutting plotter. (c) Base-keypad on the acrylic substrate. (d) Applying the first layer of ink coating. (e) Testing the electrical resistance of the circuit. (f) Successful fabrication of a lock security keypad with as synthesized ink.

6. Conclusions
In conclusion, this study successfully synthesized conductive silver ink via the chemical reduction of silver nitrate and sodium acetate and subsequently evaluated its performance across various ratios of silver acetate and ammonium solution. Through meticulous analysis, it was determined that the (1:3 ratio) sample solution exhibited an impressive purity of 93.59% silver composition as confirmed by EDX imaging. Furthermore, the sizes of silver nanoparticles within the conductive ink were effectively reduced through the application of sonication and centrifugal force, highlighting the versatility and adaptability of the ink formulation. Importantly, the synthesized conductive silver ink demonstrated multifaceted utility across a diverse array of electronic applications, including but not limited to lock security keypads, circuitry, ribbon cables, RFID tags, soft-potentiometers, and radio frequency antenna systems such as those utilized in Bluetooth and Wi-Fi technologies. These findings underscore the potential of the developed conductive silver ink to significantly impact and enhance various electronic applications, thereby contributing to the advancement of modern technology and innovation.
Conflict of Interest
The authors declared that there is no conflict of interest.
Author Note
Sai Ko Soe and Zaw Min Hein contributed equally to this manuscript.
We thank Dr. Maung Maung Thant for his contribution to transcribing and analyzing the research data. We would also like to thank the Defence Services Academy for funding this research.
Correspondence concerning this article should be addressed to Dr. Maung Maung Thant, Department of Physics, Defence Services Academy, PyinOoLwin, Myanmar.
Author Address and Emails
Sai Ko Soe1,#, Email: kaunggy@gmail.com
Zaw Min Hein1,#,Email: zminhein42@gmail.com
Phyu Phyu Thwel4, Email: phyuphyuthwel@bit.edu.cn
Kyaw Zin Oo3, Email: kyawzinoo7290@gmail.com
Maung Aye1, Email: rdan5157@gmail.com
Ye Naing1, Email: koyenaing2009@gmail.com
Lin Htun1, Email: 11linnhtun@gmail.com
Wine Htet Soe2, Email: winw58699@gmail.com
Thida Soe2, Email: soethida621@gmail.com
Kyawt Kyawt Khaing1, Email: kyawtkyawtkhine598@gmail.com
Phyu Phyu Thant1, Email: phyuphyuthant.staffoffcier@gmail.com
Sitt Paing Myint3, Email: sittpaingmyint48@gmail.com
Maung Maung Thant3, *, Email: mgmgthant48@gmail.com
1School of Science, Harbin Institute of Technology, Shenzhen-518055, China.
2Department of Botany, Yadanabon University, Mandalay, 05011, Myanmar
3Department of Physics, Defence Services Academy, Pyin Oo Lwin, Myanmar.
4School of Materials Science and Engineering, Beijing Institute of Technology, Beijing 100081, China
References
Dearden, A. L., Smith, P. J., Shin, D. Y., Reis, N., Derby, B., & O’Brien, P. (2005). A Low Curing Temperature Silver Ink for Use in Ink-Jet Printing and Subsequent Production of Conductive Tracks. Macromolecular Rapid Communications, 26(4), 315–318. doi: 10.1002/MARC. 200400445
Grouchko, M., Kamyshny, A., Mihailescu, C. F., Anghel, D. F., & Magdassi, S. (2011). Conductive inks with a “built-in” mechanism that enables sintering at room temperature. ACS Nano, 5(4), 3354–3359. doi: 10.1021/NN2005848/SUPPL_FILE/NN2005848_ SI_001.PDF
Iravani, S., Korbekandi, H., Mirmohammadi, S. V., & Zolfaghari, B. (2014). Synthesis of silver nanoparticles: chemical, physical and biological methods. Research in Pharmaceutical Sciences, 9(6), 385. Retrieved from https://pmc.ncbi.nlm.nih.gov/articles/PMC4326978/
Islam, N., Das, M., Johan, B. A., Shah, S. S., Alzahrani, A. S., & Aziz, Md. A. (2025). Multifunctional Screen-Printed Conductive Inks: Design Principles, Performance Challenges, and Application Horizons. ACS Applied Electronic Materials. doi: 10.1021/ACSAELM. 5C01256
Jiang, H., Tang, C., Wang, Y., Mao, L., Sun, Q., Zhang, L., Song, H., Huang, F., & Zuo, C. (2021). Low content and low-temperature cured silver nanoparticles/silver ion composite ink for flexible electronic applications with robust mechanical performance. Applied Surface Science, 564, 150447. doi: 10.1016/J.APSUSC.2021.150447
Suriati, G., Mariatti, M., & Azizan, A. (2011). Effects of filler shape and size on the properties of silver filled epoxy composite for electronic applications. Journal of Materials Science: Materials in Electronics, 22(1), 56–63. doi: 10.1007/S10854-010-0082-2/FIGURES/7
Wang, W., & Gu, B. (2004). Preparation and Characterization of Silver Nanoparticles at High Concentrations. 1–14. doi: 10.1021/BK-2004-0878.CH001
Yang, L., Huang, X., Wu, H., Liang, Y., Ye, M., Liu, W., Li, F., Xu, T., & Wang, H. (2023). Silver Nanowires: From Synthesis, Growth Mechanism, Device Fabrications to Prospective Engineered Applications. Engineered Science, 23. doi: 10.30919/ES8D808

Address
E-304, Mya Kyi Ryar 2nd Street, Htan Ta Pin Special Block, Dekkina Thiri Township, Naypyitaw.
Phone
(+959) 945 404 777
(+959) 882 640 707